Earth’s atmosphere
The atmosphere of Earth makes the planet habitable and contains all of its weather.
Earth’s atmosphere composition
Air is made up of a mixture of different gases. The principal constituents of Earth’s atmosphere are:
- Nitrogen – about 78% of the atmosphere
- Oxygen – about 21%
- Argon – about 1%
As well as these, there are various trace gases at relatively low concentrations measured in parts per million (ppm; 1ppm = 0.0001%)
- Carbon dioxide – about 416 ppm
- Neon – about 18 ppm
- Helium – about 5 ppm
- Methane – about 1.8 ppm
- Krypton – about 1.1 ppm
Concentrations of carbon dioxide, as well as other greenhouse gases like methane, have been rising in recent centuries because of human activity. See our introduction to climate change for more.
In addition to the above, the atmosphere also contains water vapour at a concentration of up to 3%. However, this varies significantly over time and in different places.
While the relative concentration of most gases in the atmosphere is constant at low levels, the overall pressure falls with altitude. This is why there is less oxygen available at high altitude, because the atmospheric pressure is so much lower.
Earth’s atmosphere layers
The Earth’s atmosphere is divided into several layers, defined according to changes in temperature with altitude. The boundaries between the layers are not precisely fixed, but can vary somewhat.
Troposphere
This is the lowest level of the atmosphere, extending from the surface up to between about 9 km and 17 km altitude, depending on latitude and weather conditions. The troposphere is the densest atmospheric layer because of the weight of the overlying layers, containing about 75% of the mass of the entire atmosphere.
Nearly all weather takes place in the troposphere. 99% of all the water vapour in the atmosphere is found in the troposphere, hence almost all clouds form here too. This is also the layer in which most aeroplanes fly.
Air in the troposphere becomes colder at higher altitudes up to its upper boundary, called the tropopause.
Stratosphere
Above the tropopause we find the stratosphere, which extends up to about 50 to 55 km altitude. Air temperatures in the stratosphere increase with altitude, unlike in the troposphere.
The stratosphere contains the ozone layer, a part of the atmosphere that contains a relatively high concentration of ozone between roughly 15 to 35 km in altitude. The ozone layer absorbs ultraviolet radiation from the Sun, which would otherwise be harmful for living things on the Earth’s surface. Absorbing this radiation is what causes the stratosphere to be much warmer than the troposphere.
The air in the stratosphere is much more stable than in the troposphere, therefore we don’t see much weather in this layer. However, very cold conditions can produce nacreous clouds, which form in the lower stratosphere. Weather balloons sent to capture data on the atmosphere typically reach somewhere in the stratosphere before bursting.
Mesosphere
Pass through the top of the stratosphere and you will find yourself in the mesosphere. This layer extends up to about 80 to 85 km altitude and is marked by another change in temperature profile.
The mesosphere gets colder with altitude, much like the troposphere. The top of the mesosphere is the coldest part of the atmosphere, reaching temperatures as low as -100°C.
Even with water vapour being extremely scarce in the mesosphere, the temperature is so low that clouds of ice crystals can still form. These rare noctilucent clouds are the highest clouds found on Earth.
Thermosphere
The thermosphere is the layer above the mesosphere, although by the time we reach this altitude the idea of an atmosphere as we understand it is beginning to break down. The top of the thermosphere, the thermopause, is usually where the top of the atmosphere is defined and is found at an altitude of between 500 and 1000 km depending on location, time of day, season, and other factors.
In another temperature inversion, the temperature of the thermosphere rises with increasing altitude. However, the air in the thermosphere is so thin that temperature in the usual sense is not very useful. Temperature is a measure of how much energy molecules have, but this can only be measured if one of these molecules hits something. Therefore, although air molecules in the thermosphere might have energy equivalent to a temperature of over 1,500°C, there are so few of them that the thermosphere would not feel hot to the touch.
The line marking the boundary of outer space is found in the lower thermosphere, at 100 km altitude. Aurorae typically occur in the thermosphere, and you will also find many satellites orbiting here, including the International Space Station.
Exosphere
Above the thermosphere, the final layer of Earth’s atmosphere is the exosphere. This doesn’t really have a well-defined upper limit, as it merges with interplanetary space where gases can escape Earth’s gravitational pull.
Although molecules in the exosphere can have very high energy, they may travel hundreds of kilometres before they hit anything, even another molecule. Hence, the exosphere would not feel hot, much like the thermosphere.
At the top of the exosphere, gas molecules are constantly migrating in and out of the solar wind and the planet's magnetic field, as Earth’s gravitational pull is so weak at this altitude.
The only other things to be found in the exosphere are artificial satellites and occasional aurorae. Beyond the exosphere, we leave Earth’s atmosphere behind and enter the interplanetary medium, dominated by the stream of particles emitted by the Sun.
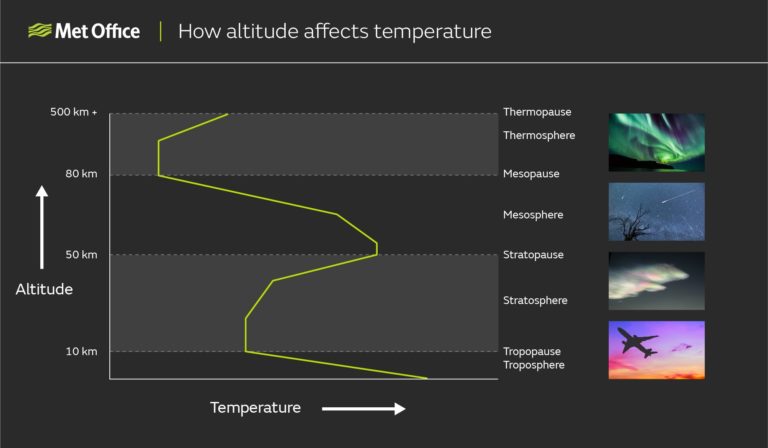
This chart illustrates how the temperature of the atmosphere goes either up or down with increasing altitude, with changes in the direction of this trend marking the boundaries between the different layers of the atmosphere.
Earth’s early atmosphere
The Earth’s atmosphere has not always had the same composition as today. Early in its history, Earth’s atmosphere would have been quite alien to us.
After the Earth formed it was subject to bombardment by asteroids and other celestial bodies. Once this ended and the planet stabilised, the first proper atmosphere would have been dominated by gases from volcanoes and other geological activity.
The early atmosphere is thought to have consisted largely of nitrogen and carbon dioxide. Carbon dioxide concentrations were many times higher than today, while oxygen was practically absent.
The high levels of carbon dioxide may have allowed some parts of the carbon cycle, such as rock weathering and oceanic gas exchange, to begin as early as 4 billion years ago.
The evolution of living things had a profound influence on the atmosphere, particularly cyanobacteria. These microbes were the first organisms to create oxygen through photosynthesis, in the process also absorbing carbon dioxide.
Initially, most of this oxygen was absorbed into various reservoirs, largely sediments. Only a small amount of oxygen was present in the atmosphere, but this was still enough to contribute to a major extinction of many microbe species that had evolved without oxygen, as oxygen was toxic to them. This Great Oxygenation Event took place between about 2.4 and 2 billion years ago.
Eventually these reservoirs were filled, and atmospheric oxygen levels began to build from about 850 million years ago.


